Investors
- Home
- Investors
Investors
For Investors & Potential Customers
MicroProtein Technologies (MPT) is an emerging Contract Manufacturing Organization (CMO) dedicated to providing custom protein production services for bio-pharmaceutical, enzyme and plasmid DNA industries, as well as academic research labs.
MicroProtein Technologies’ mission is to become a leading CMO that provides high quality and cost effective protein production services that are recognized throughout the bio-pharmaceutical industry.
MicroProtein Technologies’ strategy is to provide protein production services through its patented platform technology: MPTxpress.
MPT’s platform technology has a broad applicability and can be applied to almost any recombinant proteins expressed in Escherichia coli. Utilizing a simple yet efficient platform technology developed by MPT, a protein of interest can be easily produced, correctly folded, and purified from the host with minimal up-stream and down-stream processing requirements. With our platform it is feasible to significantly limit the cost and time of production.
Since the late 1990s, the overall CMO/CDMO market has grown significantly to around $750 billion.
Investors
Let’s See Our Latest Project

Investment Management
Lorem ipsum dolor sit amet, consec adipiscing elit, sed do eiusmod tempor incididunt ut labore

Money Calculations
Lorem ipsum dolor sit amet, consec adipiscing elit, sed do eiusmod tempor incididunt ut labore

Financial Planning
Lorem ipsum dolor sit amet, consec adipiscing elit, sed do eiusmod tempor incididunt ut labore

Data Entry
Lorem ipsum dolor sit amet, consec adipiscing elit, sed do eiusmod tempor incididunt ut labore
Testimonials
We Are Trusted Over 16+ Countries Worldwide

Entrepreneur
Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua Ut enim ad minim veniam

CEO Bciaga
Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua Ut enim ad minim veniam

Businessman
Investors
$750 billion Market by 2028
The R&D pipeline for new drugs has changed considerably over the past decade. Once dominated by big pharma and small molecules, today’s pipeline is increasingly populated with small and emerging biotech companies developing large molecules, or biologics, which now make up nearly 40% of the active R&D pipeline globally.
As scientific advancement in biopharmaceuticals continues to rise, so will the market. The global biological market size is expected to reach $750 billion in 2028 — up from the $285 billion biologics revenues seen in 2020. This rapid and continual growth has created a demand for therapeutics, expanding indications for biologics, and the growing portfolios of biologics. Biologics are mainly used for treatment of conditions such as cancer, anemia and other autoimmune diseases and the current success rate is encouraging bio-pharmaceutical players to invest in research and development of biologics on large scale. While these projections are good news for patients and for makers of bio therapies, growth always comes with new challenges. In this case, the industry must learn how to deploy efficient, flexible-modular manufacturing technologies that can respond to upstream and downstream variables. These variables include the proliferation of new biologics, rapid shifts in volumetric requirements, and improvements in cell and bacterial culture strategies.
MPTxpress process is well positioned to offer tangible benefits such as modular batch size, lower operating cost, and faster turnaround and reduced capital investment. We draw on our manufacturing and quality expertise to design processes that optimize product yields.
Brokerage
Erossed seronite mporise aliesente in consece erroe desonsed resonse.
Mutual Fund
Consequat erossed ones seonempor innsecterdum phasellus laoreet vol...
Insurance
Intege consequat erossed seroni tempor esentesera rutrum tempor felis....
Valuable Services
Mtege consequat erossed eronitem aliese sedesei egestas tellusedrty...
CMO
cGMP Biologic Development and Manufacturing
MPTxpress Process Development Services and Capabilities
MPT routinely performs upstream and downstream development and optimization studies for our clients, is a critical step towards efficient design of cGMP-compliant manufacturing processes.
MPT has experience with modular MPTxpress of E.coli. MPTxpress processes are demonstrated in process development and scaled-up for cGMP in our facility.
Protein Purification and Bulk Fill
MPT has experience with a wide range of protein purification capabilities to meet client’s cGMP production needs. Protein purification areas at MPT are segregated and customized with equipment to meet the requirements of client’s process. Protein purification personnel at MPT are experts with protein refolding, multiple chromatography techniques, tangential flow filtration, lyophilization, and bulk drug substance filling.
Microbial Cell Banking
MPT Biologics manufactures and cryo-stores Working and Master Cell Banks to support current and future upstream and downstream requirements. Microbial Cell Banks are cryo-stored in the vapor phase of liquid nitrogen freezers. MPT manages and coordinates all required testing per ICH and FDA guidelines.
CMO
MPTxpress Technology
The majority of recombinant proteins currently available are produced from bacteria. The production process comprises of large-scale fermentation to yield high volumes of bacteria. Efficient production of recombinant proteins in bacteria, both at the diagnostic and commercial level, remains a major problem. Not all recombinant proteins can be expressed to high level with a single technology.
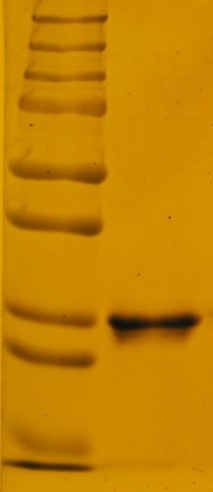
There is a need for a technology that will address major challenges of biologically active protein production. MicroProtein Technologies has generated data (Proof of Concept) to demonstrate that solid media can be used in place of the standard liquid fermentation process to produce a high yield of biologically active recombinant proteins in bacteria. MicroProtein Technologies will be the first company to produce recombinant proteins on a commercial scale without employing conventional fermentation process.
The core innovation involves creating a process that is assured to perform with the same efficacy in small scale as well as in industrial large-scale production. This process eliminates costly steps used today in recombinant protein production. This platform technology is designed to service the needs of bio-pharmaceutical companies and academic research laboratories by reducing the cost and time for the production of commercially important protein drug products.
CRO and CDMO projects performed at MicroProtein Technologies Inc.
Our Clients
We Serve All Over The World
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Donec vel dignissim lectus. Pellentesque et scelerisque nunc. Sed vel ipsum auctor, iaculis arcu quis, posuere duiuis condimentum.






Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua Ut enim ad minim veniam